Mirdametinib in Histiocytic Disorders
Phase II trial. All ages (2+), all histiocytoses including ECD. MEK inhibitor with strong preclinical rationale across MAPK-driven conditions.
Understanding Erdheim-Chester Disease.
There’s a particular kind of patient that Erdheim-Chester Disease tends to create. Because ECD is so rare, most people who receive this diagnosis quickly realize they are going to have to become deeply, almost professionally, informed about their own condition.
Your GP likely hasn’t seen it. Your local oncologist may be consulting literature they’ve never had reason to read before. The path to diagnosis was probably long, strange, and full of wrong answers. And somewhere along that path, you became an expert — not because you wanted to, but because you had no choice.
That expertise deserves a home. A place where what you’ve learned through years of navigating this condition can help someone who was just diagnosed last week.
ECD sits at a strange intersection — classified as a histiocytic neoplasm (a slow-growing blood cancer), capable of affecting almost any organ system, with treatment protocols still being shaped in real time by the patients and researchers living and working with it. The community that exists around ECD is small but remarkably informed, and remarkably generous with that information.
I want this to be a space that reflects that generosity. My condition is Hajdu-Cheney Syndrome — another condition so rare that most doctors encounter it only in textbooks. You deserve a space that’s yours, not scattered across corners of the internet.
Classified by the WHO in 2016 as a histiocytic neoplasm — a slow-growing blood cancer driven by somatic mutations in immune cells. Not inherited. Not contagious. Not passed to children.
ECD can infiltrate bones, heart, blood vessels, kidneys, lungs, brain, eyes, and skin. The “hairy kidney” and “coated aorta” on imaging scans are hallmark signatures your radiologist may recognize.
Over 80–95% of ECD patients carry mutations in the MAPK/ERK signaling pathway. Once you know your mutation status, treatment becomes targeted and dramatically more effective.
Think of the MAPK/ERK pathway (also called the RAS-RAF-MEK-ERK pathway) as your cells’ internal growth-control switchboard. It is a precise chain of proteins that carries signals from receptors on the cell surface all the way to the cell’s nucleus.
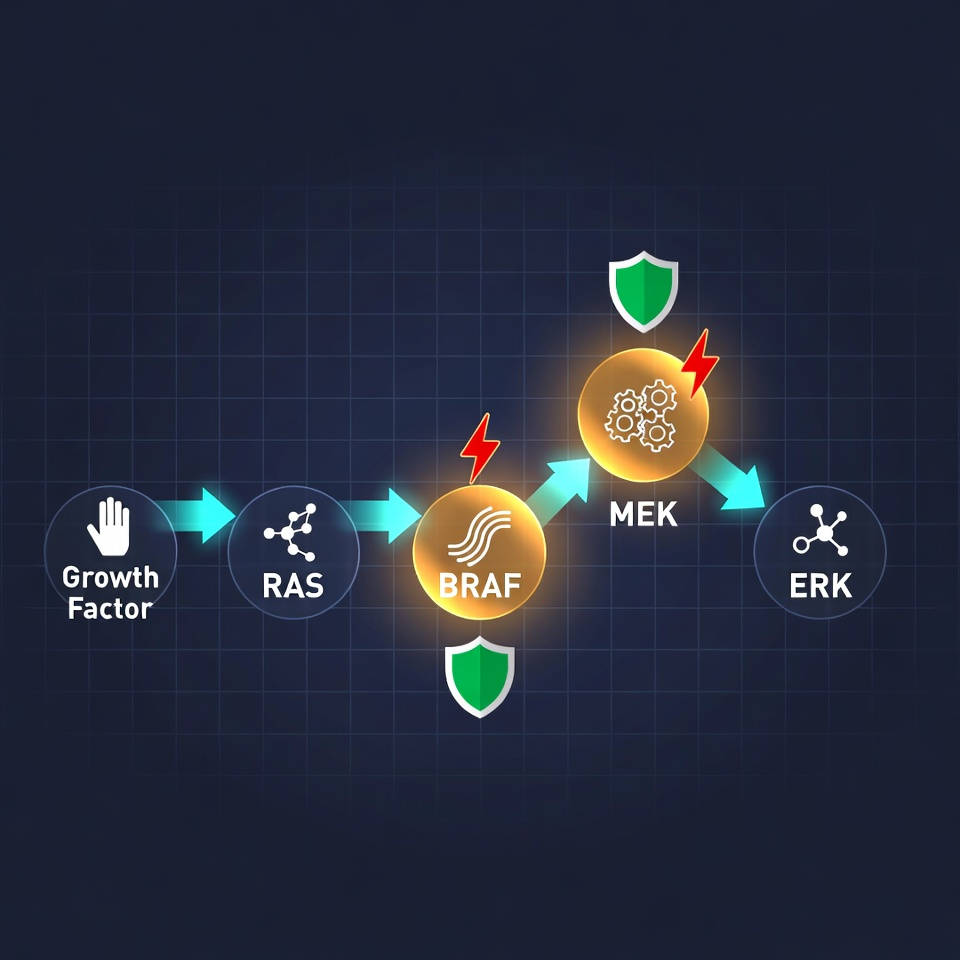
In ECD, somatic mutations (acquired during life, NOT inherited) create a “stuck-ON” switch. The pathway fires constantly, even without any external signal. This drives your myeloid-derived histiocytes — normal immune cells — to proliferate uncontrollably, become lipid-laden “foamy” cells, and invade tissues, causing the hallmark sclerosis, fibrosis, and organ damage you already know too well.
The 2012 discovery of these mutations (starting with BRAF V600E) was the turning point that proved ECD is neoplastic, not “just inflammatory.” Once you know your mutation status, treatment becomes targeted.
Directly block the mutant BRAF protein. Vemurafenib was FDA-approved for ECD in 2017 — the first targeted therapy for this disease.
Work downstream across almost the entire pathway. Effective for patients without BRAF V600E who carry MAP2K1 or other MAPK alterations.
BRAF inhibitor paired with a MEK inhibitor for deeper pathway suppression. Especially considered for aggressive or treatment-resistant disease.
The next frontier: blocking ERK directly for patients who develop resistance to upstream inhibitors. Currently in clinical trials.
What patients report: Many see PET-avid lesions shrink within weeks to months. Bone pain eases. Exophthalmos improves. “Coated aorta” and “hairy kidney” stabilize. Therapy is usually lifelong, but often at much lower doses than used in melanoma. Side effects — rash, joint pain, fatigue, skin sensitivity — are manageable and frequently improve with dose adjustment. ECD patients may be particularly sensitive to standard dosing, so dose reductions are common and do not diminish response.
Phase II trial. All ages (2+), all histiocytoses including ECD. MEK inhibitor with strong preclinical rationale across MAPK-driven conditions.
Phase II at Memorial Sloan Kettering Cancer Center and partners. Targets ERK directly for resistant or refractory disease.
The ECD Global Alliance Patient Navigator will match you personally — often securing trial access, free drug programs, or referral to an ECD Care Center. Don’t navigate this alone.
Jakob Erdheim and William Chester describe ECD as “lipoid granulomatosis” in Vienna.
Dr. Ronald Jaffe coins the term after reporting a similar case.
Researchers discover recurrent BRAF V600E mutations — proving ECD is a clonal neoplasm. Targeted therapy begins.
WHO classifies ECD as a histiocytic neoplasm, legitimizing research funding and insurance coverage.
First targeted therapy approved for ECD, based on the VE-BASKET trial.
MEK inhibitors show efficacy. Mayo Clinic and MSKCC publish consensus guidelines.
ECDGA hosts Patient & Family Gathering in Barcelona. Mirdametinib receives first FDA approval (NF1).
Registration open for the 2026 Patient & Family Gathering in Birmingham, Alabama.
I spent four years being told nothing was wrong. Then another two being told they didn’t know what was wrong. When I finally got the diagnosis, I wasn’t relieved — I was furious. And then I got to work.
The day I found the ECD community online was the first time I didn’t feel like I was making this up. These people understood every scan, every setback, every small victory.
Six months on vemurafenib and my bone pain was gone. My PET scan cleared. I went from planning my funeral to planning my daughter’s graduation.
My oncologist had never seen a case. I printed out the consensus guidelines and brought them to every appointment. We learned together.
The MAPK/ERK pathway that once drove your disease is now the exact roadmap to controlling it. You already mastered the hardest part — surviving the diagnostic odyssey and becoming the expert your care team needed.
Connect with the ECD Global Alliance (opens in new tab)Share your mutation story, your before-and-after scans, your side-effect insights, your plans for the 2026 Birmingham Gathering.
#BareYourRareECD
You are not alone. You are home.
All information verified February 2026 from the ECD Global Alliance, Blood consensus guidelines (2020/updated), recent 2025 publications, ClinicalTrials.gov, Histiocytosis Association, and patient-reported sources.
Last reviewed:
Community Voices
Stories and insights from the ECD community will appear here as they are submitted and reviewed. Be the first to share yours.
No stories yet — yours could be the first.